PEEK Interference Screw – Biocompatible Fixation for ACL and Soft Tissue Reconstruction
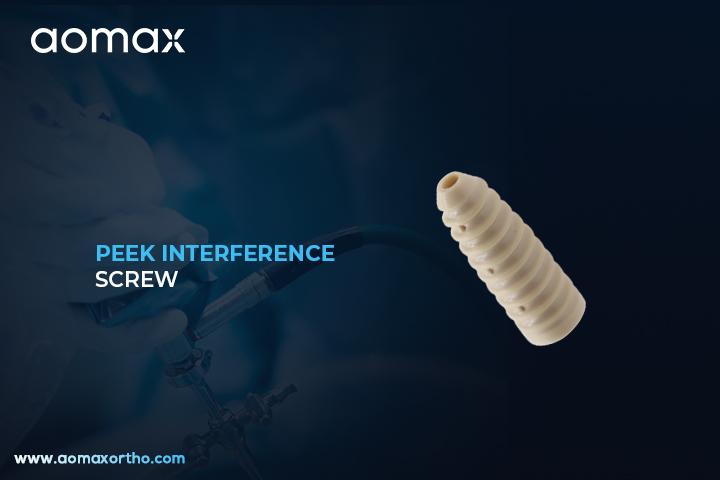
PEEK Interference Screw
The PEEK Interference Screw is a medical implant used to fix soft tissue grafts to bone, especially during ligament reconstruction procedures like ACL or PCL surgery. It is made from PEEK (Polyetheretherketone), a high-strength, biocompatible polymer. This screw offers strong fixation while minimizing potential imaging interference due to its radiolucent nature.
Product History
Interference screws have been in use since the late 1980s for sports medicine and orthopedic surgeries. Initially made from metal, advancements led to the development of bioabsorbable and polymer-based materials. PEEK became popular due to its strength, biocompatibility, and non-reactive properties. Over the years, PEEK Interference Screws have gained widespread clinical acceptance in knee and shoulder ligament reconstructions.
Trusted Support for ACL and Soft Tissue Reconstruction
Aomax Ortho offers PEEK Interference Screws that provide secure graft fixation with minimal risk of reaction or imaging distortion. These screws are made from high-performance PEEK material, ensuring long-term stability and biocompatibility. Aomax Ortho designs them in various sizes to accommodate different graft types and surgical preferences. The threaded design ensures optimal compression between the graft and bone tunnel. This results in enhanced healing and faster patient recovery.
Surgeries Related to Product
- ACL (Anterior Cruciate Ligament) Reconstruction
- PCL (Posterior Cruciate Ligament) Reconstruction
- MCL (Medial Collateral Ligament) Repair
- Meniscal Root Repair
- Shoulder Ligament Reconstruction
- Ankle Ligament Reconstruction
Instructions for Use
- Choose appropriate screw size based on graft and tunnel dimensions
- Insert the graft into the prepared bone tunnel
- Align the screw with the tunnel and graft
- Insert screw carefully to avoid graft damage
- Confirm fixation via tactile feedback and stability
Clinical Benefits
- Radiolucent material allows clear post-op imaging
- High pull-out strength ensures secure graft fixation
- Biocompatible and non-metallic, reducing the risk of allergic reactions
- Available in multiple sizes to match different surgical requirements
- Does not interfere with MRI or CT imaging
- Promotes graft-to-bone healing through stable compression
Product Also Known As
PEEK ACL Screw, PEEK Ligament Fixation Screw, Polymer Interference Screw, Radiolucent Interference Screw, Biocompatible PEEK Screw, Orthopedic PEEK Fixation Screw
Frequently Used Product Combination
- ACL/PCL Reconstruction Instruments Set
- Arthroscopic Cannulas and Passers
- Femoral Endo Button (Titanium or PEEK)
- Tibial Fixation Screw
- Suture Anchors (for soft tissue support)
- Bone Tunnels Dilator Set
Market Demand and Global Trends
PEEK Interference Screws are in high demand globally due to the rise in sports injuries and ligament reconstruction surgeries. Surgeons prefer PEEK over metallic screws for its compatibility with diagnostic imaging and long-term biostability. Developed regions like the US and Europe show strong adoption, while emerging markets in Asia-Pacific are rapidly expanding. The trend towards minimally invasive and arthroscopic procedures continues to boost its global usage.
Aomax PEEK Interference Screw Description
Aomax Ortho is a leading manufacturer of PEEK Interference Screws designed for advanced orthopedic fixation with high biocompatibility, offering reliable performance in ligament reconstruction procedures.
Get Connected:
+91 98989 50530 | exports@aomaxortho.com | www.aomaxortho.com
Anterior Cervical Screw – Secure Fixation for Cervical Spine Fusion and Stability
Anterior Cervical Screw (ACL Screw)
The Anterior Cervical Screw (ACL Screw) is a spinal implant used for anterior cervical spine stabilization. It is inserted into the cervical vertebrae to secure plates during fusion procedures, especially in cases of disc degeneration, trauma, or instability. Made from biocompatible titanium, it provides strong fixation and long-term spinal support.
Product History
Anterior cervical fusion has become a widely accepted surgical method for treating cervical spine disorders. Over time, implant systems evolved from basic fixation methods to precision-engineered plates and screws. The ACL-type anterior cervical screw was developed to improve grip, prevent back-out, and enhance fusion outcomes. Today, it is a key component in anterior cervical plating systems used worldwide.
Engineered for Secure, Low-Profile Spinal Stability
Aomax Ortho offers Anterior Cervical Screws (ACL Screws) specifically designed for use in anterior cervical plate systems. These screws ensure solid fixation to the cervical vertebrae and provide the necessary mechanical support for spinal fusion. Aomax Ortho manufactures them using medical-grade titanium with self-tapping threads for ease of insertion and stability. The screw head design is compatible with locking and non-locking plates to meet surgical preferences. These screws are widely used in trauma, degenerative disc disease, and spinal deformity corrections.
Surgeries Related to Product
- Anterior Cervical Discectomy and Fusion (ACDF)
- Cervical Spine Trauma Surgery
- Cervical Degenerative Disc Disease Treatment
- Cervical Vertebral Fracture Fixation
- Spinal Deformity Correction Procedures
Instructions for Use
- Position the anterior cervical plate over the prepared vertebral bodies
- Align the screw hole with the pilot hole drilled in the bone
- Select the appropriate screw length based on bone depth
- Insert the ACL screw using a compatible screwdriver
- Lock the screw head into the plate if using a locking system
Clinical Benefits
- Provides rigid fixation for anterior cervical fusion
- Compatible with locking plate systems to prevent screw back-out
- Self-tapping threads reduce surgical time
- Titanium construction ensures biocompatibility and imaging clarity
- Designed to match cervical anatomy and minimize soft tissue irritation
Product Also Known As
ACL Cervical Screw, Anterior Cervical Fusion Screw, Cervical Plate Fixation Screw, Spine Locking Screw, Cervical Vertebral Screw, Cervical Plate Screw, Titanium Cervical Screw, Anterior Cervical Locking Screw, Spinal Fixation Screw, Self-Tapping Cervical Screw
Frequently Used Product Combination
- Anterior Cervical Plate (ACP)
- Cervical Distractor Set
- Titanium Cage or Spacer
- Surgical Screwdriver (Hex or Torx Type)
- Cervical Drill Guide
- Fluoroscopic Imaging System
Market Demand and Global Trends
With the increasing incidence of cervical disc degeneration, trauma, and spinal instability, the demand for anterior cervical fusion systems continues to rise. Surgeons prefer ACL-type screws for their reliable locking, precision threading, and compatibility with advanced plate systems. Titanium screws remain the standard due to excellent imaging compatibility and durability. The product is seeing strong adoption in hospitals and spine centres across North America, Asia-Pacific, and Europe.
Aomax Anterior Cervical Screw (ACL Screw) Description
Aomax Ortho is a trusted manufacturer of Anterior Cervical Screws (ACL Screws), delivering high-precision spinal fixation solutions that enhance stability, fusion success, and patient recovery in cervical spine surgeries.
Get Connected:
+91 98989 50530 | exports@aomaxortho.com | www.aomaxortho.com
Titanium Femoral Button – Reliable Cortical Fixation for ACL and Soft Tissue Reconstruction
Femoral Button (Titanium)
The Femoral Button (Titanium) is an orthopedic implant used for cortical fixation of soft tissue grafts in ligament reconstruction surgeries, especially ACL repair. It provides secure anchorage on the femoral side by suspending the graft from the outer cortex of the femur. Made from surgical-grade titanium, it ensures strength, biocompatibility, and long-term stability.
Product History
Fixation devices for ACL and PCL reconstruction have evolved to enhance graft security and minimize surgical complexity. Femoral buttons were introduced to replace bulky screws and allow less invasive femoral fixation through cortical suspension. Titanium versions became popular due to their durability, low profile, and compatibility with biological tissues. Today, femoral buttons are a standard choice in sports medicine procedures worldwide.
Engineered for Fast Deployment and Lasting Stability
Aomax Ortho offers Femoral Button (Titanium) designed for strong and reliable cortical fixation in ACL and PCL reconstruction. This titanium implant provides high pull-out strength and ensures consistent tension on the soft tissue graft. Aomax Ortho ensures a smooth, low-profile button design that simplifies deployment and minimizes irritation to surrounding tissue. The button passes through the femoral tunnel and flips to lock securely against the cortex. It supports fast recovery and secure healing in both primary and revision ligament surgeries.
Surgeries Related to Product
- ACL Reconstruction
- PCL Reconstruction
- Multiligament Knee Repair
- Revision Ligament Reconstruction
- Soft Tissue Graft Fixation in the Femur
Instructions for Use
- Create a femoral tunnel using an appropriate drill guide
- Pass the graft through the tunnel with the button attached
- Pull the graft until the button exits the femoral cortex
- Flip the button and confirm it is locked on the outer cortex
- Adjust tension as needed before securing the tibial end
Clinical Benefits
- High-strength titanium offers long-term graft support
- Secure cortical suspension improves graft stability
- Minimally invasive implantation preserves bone structure
- Low-profile design reduces soft tissue irritation
- Ideal for arthroscopic and open ligament reconstruction
Product Also Known As
Titanium Femoral Fixation Button, ACL Femoral Button, Cortical Suspension Button, Titanium Graft Fixation Button, Femoral Endobutton, Ligament Femoral Anchor, Titanium ACL Fixation Button, Femoral Tunnel Button, Graft Suspension Device, Titanium Cortical Fixation Implant
Frequently Used Product Combination
- Tibial Interference Screw
- ACL/PCL Graft Tensioner
- Femoral Drill Guide
- Endoscopic Cannula
- Suture Passing Device
- Arthroscopy Instruments Set
Market Demand and Global Trends
The Femoral Button (Titanium) is in high demand due to its reliable performance in ligament reconstruction and growing global interest in minimally invasive sports surgeries. Surgeons prefer cortical suspension systems for their simplicity, reduced tunnel widening, and fast recovery outcomes. Titanium remains a gold standard due to its strength and imaging compatibility. With the increase in ACL injuries and sports-related trauma, usage is expanding across orthopedic centers in North America, Europe, and Asia.
Aomax Femoral Button (Titanium) Description
Aomax Ortho is a leading manufacturer of Femoral Button (Titanium), offering precise, durable, and surgeon-friendly solutions for secure graft fixation in advanced orthopedic ligament reconstruction procedures.
Get Connected:
+91 98989 50530 | exports@aomaxortho.com | www.aomaxortho.com
Endo Button Knotless Rope with Brace – Implant-Free Soft Tissue Fixation for ACL Reconstruction
Endo Button Knotless Rope with Brace
The Endo Button Knotless Rope with Brace is a high-performance orthopedic implant designed for soft tissue fixation, particularly in ACL and PCL ligament reconstruction surgeries. It combines a cortical fixation button with a pre-assembled rope system that eliminates the need for surgical knots, improving consistency and reducing operative time. The addition of an integrated brace allows precise tension adjustment, offering enhanced control and stability during graft placement and healing.
Product History
Conventional button fixation systems often required manual knot tying, which increased surgical time and complexity. To address this, knotless technology was developed—allowing quicker, more secure fixation with minimal variability. The addition of a brace element provided surgeons better control over graft tension and stabilization. Over the years, this system has become widely adopted in sports medicine and ligament surgeries.

Engineered for Strong, Adjustable Fixation Without the Knot
Aomax Ortho presents the Endo Button Knotless Rope with Brace, designed for precise and reliable cortical fixation in ACL and PCL reconstruction. This system eliminates the need for suture knots, reducing the risk of loosening and saving surgical time. Aomax Ortho ensures optimal rope strength and button design to maintain fixation during early rehabilitation. The integrated brace allows controlled tensioning, improving surgical outcomes and patient stability. It is ideal for both primary and revision ligament repair surgeries.
Surgeries Related to Product
- ACL Reconstruction
- PCL Reconstruction
- Multiligament Knee Reconstruction
- Revision Ligament Repair
- Arthroscopic Ligament Fixation
Instructions for Use
- Drill the femoral or tibial tunnel as per surgical plan
- Insert graft and pass the rope through the tunnel
- Deploy the Endo Button on the cortical surface
- Adjust tension using the integrated brace mechanism
- Confirm fixation before closing the incision
Clinical Benefits
- Eliminates knot-related complications
- Ensures fast and reproducible fixation
- Integrated brace supports controlled tension adjustment
- Reduces surgical time and enhances graft stability
- Ideal for both open and arthroscopic procedures
Product Also Known As
Knotless Endo Button System, Ligament Fixation Button with Rope, ACL Button with Brace, Knotless Graft Fixation Device, Cortical Suspension Device with Rope, Arthroscopic Knotless Endo Button, Adjustable Loop Fixation System, Knotless Ligament Repair Button, Endo Fixation Button, Loop and Brace Fixation Implant
Frequently Used Product Combination
- Femoral or Tibial Drill Guide
- Arthroscopic Cannula
- Graft Tensioning Device
- Endoscopic Viewing System
- Interference Screw (Femoral/Tibial Side)
- Suture Passer
Market Demand and Global Trends
With a growing preference for knotless technology in sports injuries and ligament reconstructions, the demand for systems like the Endo Button Knotless Rope with Brace is rapidly increasing. Surgeons favor it for its efficiency, reduced complication rates, and consistent results. Global markets are seeing adoption in both advanced and emerging orthopedic centres. The integration of brace-based tensioning aligns with current trends in enhanced surgical control and precision fixation.
Aomax Endo Button Knotless Rope with Brace Description
Aomax Ortho is a trusted manufacturer of the Endo Button Knotless Rope with Brace, delivering innovative, knot-free fixation systems that provide secure, reliable, and tension-controlled solutions for ligament reconstruction procedures.
Get Connected:
+91 98989 50530 | exports@aomaxortho.com | www.aomaxortho.com
Tibial Screw – Strong and Stable Fixation for ACL and Tibial Tunnel Reconstruction
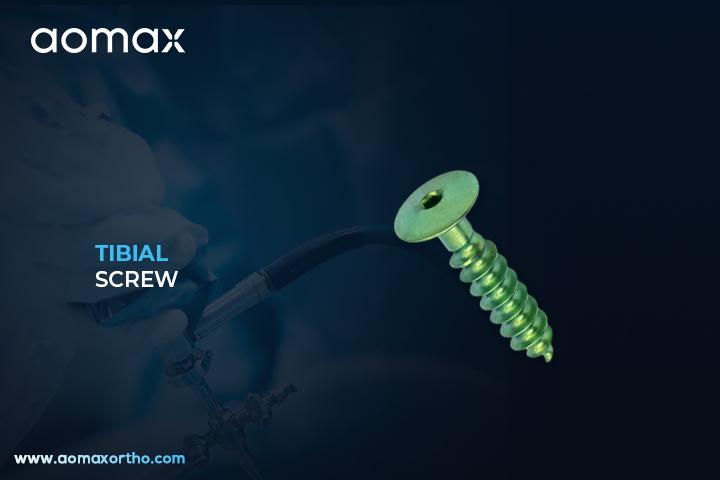
Tibial Screw
A Tibial Screw is an orthopedic implant used for the fixation of grafts in the tibial tunnel during ligament reconstruction surgeries such as ACL or PCL repair. It is designed to anchor the soft tissue graft securely against the tunnel wall. The screw plays a vital role in providing initial stability until biological healing occurs.
Product History
With the evolution of sports medicine, graft fixation in ligament reconstruction became critical for surgical success. Early fixation methods often lacked consistency, leading to the development of dedicated interference screws for the tibial side. Tibial Screws were introduced with improved thread designs and biocompatible materials for reliable fixation. Over time, they have become an essential part of arthroscopic and open knee reconstruction surgeries worldwide.

Trusted Strength for Long-Lasting Bone Support
Aomax Ortho manufactures high-performance Tibial Screws engineered for strong and secure graft fixation in ligament reconstruction surgeries. These screws are available in various sizes and materials including titanium and bioabsorbable options. Aomax Ortho ensures high thread precision and tapered tips for easy insertion and minimal graft damage. Their design provides optimal compression between the graft and tibial tunnel wall, supporting early rehabilitation. These screws are widely used in ACL, PCL, and soft tissue procedures involving tibial fixation.
Surgeries Related to Product
- ACL (Anterior Cruciate Ligament) Reconstruction
- PCL (Posterior Cruciate Ligament) Reconstruction
- Revision Ligament Reconstruction
- Patellar Tendon Graft Fixation
- Multiligament Knee Repair
- Soft Tissue Fixation in the Tibial Tunnel
Instructions for Use
- Prepare the tibial tunnel with proper sizing and alignment
- Place the graft inside the tunnel using a guide wire
- Select the correct screw size based on tunnel diameter
- Insert the screw between the graft and tunnel wall
- Advance gently to avoid graft damage and confirm fixation
Clinical Benefits
- Provides strong initial fixation for soft tissue grafts
- Promotes secure bone-to-graft healing in the tibial tunnel
- Reduces risk of graft slippage or tunnel widening
- Available in both metal and bioabsorbable options
- Compatible with various ligament reconstruction techniques
Product Also Known As
Tibial Interference Screw, ACL Tibial Screw, PCL Tibial Fixation Screw, Tibial Tunnel Screw, Ligament Fixation Screw, Graft Fixation Screw, Orthopedic Tibial Anchor, Bio Tibial Screw, Titanium Tibial Screw, Arthroscopic Tibial Screw
Frequently Used Product Combination
- Femoral Interference Screw
- ACL/PCL Drill Guide
- Graft Tensioning Device
- Tibial Tunnel Reamer
- Cannulated Screwdriver
- Graft Preparation Set
Market Demand and Global Trends
Tibial Screws are widely used in sports medicine and orthopedic trauma due to the growing number of ligament injuries and active lifestyles. Surgeons increasingly prefer precise, biomechanically stable implants that ensure faster recovery and reduced revision rates. The demand for bioabsorbable and titanium tibial screws continues to grow across North America, Europe, and Asia-Pacific regions. As arthroscopic techniques become more common, high-performance tibial fixation implants remain a standard in surgical practice.
Aomax Tibial Screw Description
Aomax Ortho is a trusted manufacturer of Tibial Screws, providing high-strength and precision-designed implants that support secure graft fixation and optimal outcomes in orthopedic ligament reconstruction surgeries.
Get Connected:
+91 98989 50530 | exports@aomaxortho.com | www.aomaxortho.com
Locking Profile Cancellous Screw – Secure Fixation for Fragile and Spongy Bone Structures
Locking Profile Cancellous Screw
A Locking Profile Cancellous Screw is an orthopedic implant used to fix fractures in cancellous (spongy) bone, especially in areas like the pelvis, proximal femur, or metaphyseal regions. It features a locking head design that secures tightly into locking plates for enhanced stability. This screw is ideal for osteoporotic bone and complex fracture patterns where strong anchorage is required.
Product History
Traditional cancellous screws offered basic bone purchase but often lacked sufficient hold in poor-quality bone. As locking plate technology developed, screws with threaded heads were introduced to lock into plate holes and form a fixed-angle construct. This evolution led to the Locking Profile Cancellous Screw—blending cancellous threading with locking capability. The design has since become essential in trauma and reconstructive surgeries, especially in elderly or osteoporotic patients.
Precision-Engineered for Stable Internal Fixation
Aomax Ortho offers Locking Profile Cancellous Screws that combine the deep-threaded grip of cancellous screws with a locking head for secure fixation. These screws are designed to lock into orthopedic plates, creating a rigid, fixed-angle construct ideal for metaphyseal and osteoporotic bone. Aomax Ortho ensures precise manufacturing from surgical-grade titanium or stainless steel for strength and biocompatibility. The screw’s design minimizes the risk of pull-out and supports early mobilization. It is an essential choice for fracture fixation in joints and load-bearing bones.
Surgeries Related to Product
- Proximal Femur Fracture Fixation
- Distal Femur or Tibia Fracture Repair
- Pelvic and Acetabular Fractures
- Osteoporotic Bone Fracture Fixation
- Periarticular Fracture Fixation
- Locking Plate System Surgeries
Instructions for Use
- Choose appropriate screw length and diameter
- Insert the screw into pre-drilled hole of the locking plate
- Engage the threaded head into the plate’s locking hole
- Advance until the screw is fully seated and locked
- Confirm placement with fluoroscopic imaging
Clinical Benefits
- Provides fixed-angle stability in osteoporotic and cancellous bone
- Prevents screw back-out and plate loosening
- Enhances fixation in metaphyseal and periarticular regions
- Reduces risk of implant failure in complex fractures
- Compatible with various locking plate systems
Product Also Known As
Locking Cancellous Bone Screw, Cancellous Locking Screw, Orthopedic Locking Screw, Threaded Head Cancellous Screw, Plate Locking Screw, Metaphyseal Locking Screw, Osteoporotic Fracture Screw, Locking Profile Trauma Screw, Locking Bone Fixation Screw, Cancellous Plate Screw
Frequently Used Product Combination
- Locking Compression Plate (LCP)
- Cancellous Bone Drill Bit
- Depth Gauge
- Screwdriver with Torque Control
- Bone Reduction Clamps
- Imaging System (C-Arm)
Market Demand and Global Trends
The global use of Locking Profile Cancellous Screws is growing rapidly, particularly due to an aging population with increasing osteoporotic fractures. Surgeons prefer locking systems for their reliable fixation and ability to reduce the need for revision surgeries. With advancements in minimally invasive fracture management, these screws are also being adapted for use in percutaneous techniques. High adoption rates are seen in trauma centers and orthopedic hospitals across North America, Europe, and Asia.
Aomax Locking Profile Cancellous Screw Description
Aomax Ortho is a trusted manufacturer of Locking Profile Cancellous Screws, providing advanced orthopedic solutions that offer reliable, fixed-angle fixation for complex and osteoporotic bone fractures.
Get Connected:
+91 98989 50530 | exports@aomaxortho.com | www.aomaxortho.com
PSLD Sheath – Precision Access Device for Endoscopic Lumbar Spine Procedures
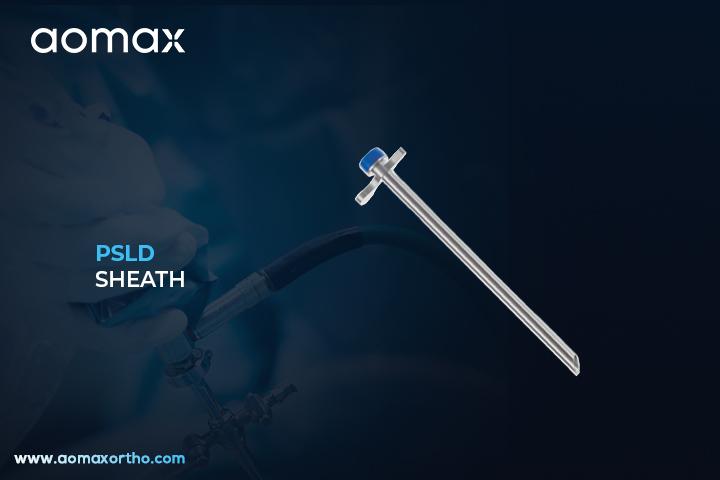
PSLD Sheath
The PSLD Sheath is a specialized surgical instrument used in Percutaneous Stenoscopic Lumbar Decompression (PSLD) procedures. It serves as a working channel to safely guide surgical tools to the lumbar spine through a minimally invasive approach. The sheath allows controlled access while protecting surrounding soft tissues during decompression.
Product History
As spinal surgery evolved toward minimally invasive methods, the need for tubular access systems became more critical. The PSLD technique, developed to treat lumbar spinal stenosis with minimal trauma, required dedicated instrumentation. PSLD Sheaths were designed to meet this demand—providing both stability and safety during access. With rising global interest in endoscopic spinal techniques, this sheath has become a key tool in advanced spinal centers.
Seamless Access for Percutaneous Spine and Ligament Procedures
Aomax Ortho offers the PSLD Sheath designed specifically for minimally invasive lumbar decompression surgeries. Engineered from high-grade stainless steel, the sheath enables precise and safe insertion of endoscopic and decompression instruments. Aomax Ortho ensures ergonomic design with optimal length and diameter for effective targeting of the stenotic area. Its atraumatic tip minimizes tissue disruption, while the locking mechanism stabilizes the sheath during procedures. This sheath supports faster recovery, reduced blood loss, and shorter hospital stays for patients.
Surgeries Related to Product
- Percutaneous Stenoscopic Lumbar Decompression (PSLD)
- Endoscopic Lumbar Foraminotomy
- Lumbar Canal Stenosis Surgery
- Lumbar Disc Herniation Decompression
- Minimally Invasive Spine Surgery (MISS)
Instructions for Use
- Prepare the patient and mark the entry point under fluoroscopy
- Make a small incision and insert the dilator sequentially
- Slide the PSLD Sheath over the final dilator to create the working channel
- Lock the sheath in position for tool insertion
- Begin decompression using endoscopic instruments through the sheath
Clinical Benefits
- Enables safe and precise access to lumbar spine
- Minimizes muscle and tissue damage
- Reduces operative time and patient recovery period
- Compatible with multiple endoscopic tools
- Ideal for outpatient spinal procedures
Product Also Known As
Endoscopic Spine Sheath, Minimally Invasive Spine Sheath, PSLD Access Tube, Lumbar Decompression Sheath, Spine Working Channel, Stenoscopic Surgery Sheath, Orthopaedic Spinal Sheath, Tubular Retractor for PSLD, Endoscopic Decompression Sheath, Lumbar Spine Access Cannula
Frequently Used Product Combination
- PSLD Endoscope
- Spine Dilator Set
- Kerrison Punch
- Bipolar RF Cautery
- Endoscopic Grasper
- Fluoroscopy Imaging System
Market Demand and Global Trends
Minimally invasive spine surgeries are on the rise globally, with PSLD gaining traction for treating lumbar spinal stenosis. The PSLD Sheath plays a critical role in these procedures, offering a safe, stable access pathway. Surgeons prefer these systems for their reduced risk, improved precision, and better patient outcomes. With the shift toward outpatient spinal care and advanced endoscopic techniques, demand for PSLD-compatible instruments continues to grow in both developed and emerging markets.
Aomax PSLD Sheath Description
Aomax Ortho is a leading manufacturer of PSLD Sheath, providing surgeons with reliable, high-precision access instruments designed to support safe, efficient, and minimally invasive spinal decompression procedures.
Get Connected:
+91 98989 50530 | exports@aomaxortho.com | www.aomaxortho.com
Titanium Interference Screw – High-Strength Fixation for Ligament and Tendon Reconstruction
Titanium Interference Screw
A Titanium Interference Screw is an orthopedic implant used to secure soft tissue grafts within bone tunnels during ligament reconstruction procedures. It provides firm, direct fixation between the graft and tunnel wall. This screw is widely used in ACL, PCL, and other joint stabilization surgeries.
Product History
Interference screws were introduced to improve graft fixation in ligament surgeries, replacing older methods like staples and sutures. Titanium was selected for its strength, biocompatibility, and radiographic visibility. Over time, design enhancements such as tapered threads and cannulated options were added. Today, Titanium Interference Screws are standard tools in arthroscopic and open joint reconstruction worldwide.
Durable Performance in Ligament Reconstruction Procedures
Aomax Ortho manufactures Titanium Interference Screws that offer secure graft fixation in ligament reconstruction surgeries. Made from surgical-grade titanium, these screws provide excellent strength, corrosion resistance, and biocompatibility. Aomax Ortho ensures optimal thread design for uniform compression between graft and bone tunnel. Their radiopacity allows easy intraoperative imaging, while the cannulated version ensures accurate placement. Ideal for ACL, PCL, and tendon fixation in knee, shoulder, and elbow surgeries.
Surgeries Related to Product
- ACL Reconstruction
- PCL Reconstruction
- Patellar Tendon Graft Fixation
- Achilles Tendon Repair
- Shoulder Instability Repair
- Elbow Ligament Reconstruction
Instructions for Use
- Prepare bone tunnel and graft as per procedure
- Insert the graft into the tunnel using a guide wire if necessary
- Select proper screw size and align with the tunnel
- Advance the screw over the graft using a driver
- Confirm fixation with imaging or tactile feedback
Clinical Benefits
- Offers strong primary fixation and promotes biological healing
- Titanium ensures long-term biocompatibility and corrosion resistance
- Radiopaque for intraoperative and post-op imaging visibility
- Cannulated options enable precise placement with guide wires
- Minimizes micromotion between graft and tunnel
Product Also Known As
ACL Interference Screw, Titanium Graft Fixation Screw, Orthopedic Tunnel Screw, Ligament Fixation Screw, PCL Titanium Screw, Cannulated Interference Screw, Sports Medicine Bone Screw, Titanium Tendon Fixation Screw, Arthroscopic Fixation Screw, Soft Tissue Tunnel Screw
Frequently Used Product Combination
- ACL Drill Guide
- Femoral and Tibial Tunnel Reamers
- Graft Tensioning Device
- Graft Preparation Board
- Guide Wire
- Arthroscopic Instruments Set
Market Demand and Global Trends
Titanium Interference Screws continue to see high demand as ligament reconstruction surgeries become more common, especially among athletes and aging populations. Their durability, compatibility with modern imaging, and consistent fixation results drive adoption globally. Surgeons favor titanium over bioabsorbable screws for long-term stability and strength in high-stress joints. Asia-Pacific and Latin America markets are also expanding rapidly due to increased sports injury awareness and surgical infrastructure development.
Aomax Titanium Interference Screw Description
Aomax Ortho is a leading manufacturer of Titanium Interference Screws, delivering high-strength, precise fixation solutions for secure graft placement in orthopedic ligament reconstruction and joint stabilization procedures.
Get Connected:
+91 98989 50530 | exports@aomaxortho.com | www.aomaxortho.com
Spiked Washer – Secure Bone-to-Bone Fixation for Orthopedic and Trauma Surgeries
Spiked Washer
A Spiked Washer is a specialized orthopedic implant used to provide additional fixation and prevent screw head migration in bone surgeries. It features small spikes on its underside that grip the bone surface securely. This washer is commonly used in trauma and reconstructive surgeries, especially where bone quality is poor or additional compression is needed.
Product History
Standard washers were initially flat and often failed to maintain grip under stress or in osteoporotic bone. Spiked Washers were developed to address this issue by incorporating pointed projections that penetrate the bone slightly to increase surface hold. Over time, they became essential in high-load fixation areas such as pelvis, ankle, and shoulder surgeries. Surgeons now prefer spiked designs for added stability and reduced implant slippage.
Enhanced Grip for Secure Bone Fixation
Aomax Ortho provides high-precision Spiked Washers designed to enhance screw fixation in orthopedic procedures. Manufactured from biocompatible stainless steel or titanium, these washers feature a contoured surface with sharp micro-spikes for superior grip on the bone. Aomax Ortho ensures consistent quality and compatibility with standard screw systems used in trauma implants. They are especially effective in osteoporotic or soft bone cases, providing improved distribution of load and reduced implant loosening. The washer plays a key role in maintaining construct stability under dynamic conditions.
Surgeries Related to Product
- Pelvic Fracture Fixation
- Shoulder Fracture Surgery
- Ankle Fracture Fixation
- Tibial Plateau Fixation
- Revision Trauma Surgeries
- Osteoporotic Bone Fixation
Instructions for Use
- Position the spiked washer over the predrilled screw hole
- Insert the screw through the washer into the bone
- Tighten the screw to allow spikes to engage with bone surface
- Ensure full seating of the washer without overtightening
- Confirm secure fixation with imaging or direct inspection
Clinical Benefits
- Enhances screw fixation in osteoporotic or Cancellous bone
- Prevents screw head migration and loss of compression
- Provides increased surface grip and stability
- Distributes load evenly across the bone surface
- Compatible with multiple types of orthopedic screws
Product Also Known As
Orthopedic Spiked Washer, Bone Grip Washer, Serrated Washer, Locking Screw Washer, Anti-Migration Washer, Spiked Lock Washer, Bone Anchor Washer, Trauma Fixation Washer, Screw Stabilizing Washer, Spiked Support Plate
Frequently Used Product Combination
- Cancellous Screws
- Cortical Screws
- Orthopedic Plates (Pelvic, Clavicle, Ankle)
- Bone Drill and Depth Gauge
- Reduction Forceps
- Imaging System (C-Arm)
Market Demand and Global Trends
With an aging global population and rising cases of osteoporotic fractures, the demand for spiked washers is increasing rapidly. Their role in improving screw retention and reducing hardware failure is driving adoption in both trauma and reconstructive surgery. The global orthopedic implant market is embracing these specialized devices for their ability to provide added fixation in challenging anatomical and bone-quality scenarios. Surgeons worldwide are incorporating spiked washers into standard trauma kits to ensure optimal outcomes.
Aomax Spiked Washer Description
Aomax Ortho is a leading manufacturer of Spiked Washers, delivering reliable, high-grip fixation solutions that enhance orthopedic screw stability in trauma, reconstruction, and osteoporotic bone surgeries.
Get Connected:
+91 98989 50530 | exports@aomaxortho.com | www.aomaxortho.com
No Button Knotless Rope – Simplified Soft Tissue Fixation without Implants
No Button Knotless Rope
The No Button Knotless Rope is an advanced orthopedic implant used for soft tissue graft fixation without the need for tying knots or using a fixation button. It enables strong, tension able fixation in ligament reconstruction surgeries. This system is especially useful in procedures like ACL or PCL repairs where simplicity, strength, and speed are critical.
Product History
Traditional fixation methods required either suture knots or metallic buttons to anchor soft tissue grafts, which could be time-consuming and prone to complications. Knotless systems were developed to eliminate knot-tying errors while improving consistency. The No Button Knotless Rope is the next evolution, offering secure fixation with a streamlined, button-free design. It has quickly gained popularity in sports medicine and minimally invasive procedures.
Knot-Free Technology for Streamlined Soft Tissue Repair
Aomax Ortho offers the No Button Knotless Rope, a specialized implant that delivers secure graft fixation without the need for cortical buttons or suture knots. The system utilizes a looped, tension able rope made of high-strength, biocompatible fiber. Aomax Ortho ensures precision control and simplified deployment for consistent outcomes in soft tissue repair. It reduces surgical time and eliminates knot-related complications, making it ideal for both primary and revision ligament surgeries. This product is especially suited for surgeons who prefer a low-profile, all-suture fixation method.
Surgeries Related to Product
- ACL Reconstruction
- PCL Reconstruction
- Revision Ligament Surgeries
- Meniscal Root Repair
- Multi-ligament Knee Procedures
- Arthroscopic Graft Fixation
Instructions for Use
- Prepare the bone tunnel and pass the graft through
- Loop the rope through the graft as directed
- Insert and tension the rope using the dedicated delivery system
- Ensure proper graft tension and lock the system in place
- Cut excess rope and verify final positioning
Clinical Benefits
- Eliminates the need for cortical buttons and knot tying
- Offers strong, adjustable, and low-profile graft fixation
- Reduces surgical time and minimizes hardware complications
- Suitable for both arthroscopic and minimally invasive procedures
- Improves consistency and reproducibility in ligament reconstruction
Product Also Known As
Knotless Graft Rope, Button less Fixation Loop, No Button Ligament Rope, Knotless Suture Loop, All-Suture Graft Fixation Device, Button-Free Soft Tissue Fixation, No-Button ACL Rope, Orthopedic Loop System, Tension able Knotless Implant, Knotless Suspension Fixation
Frequently Used Product Combination
- ACL Drill Guide
- Graft Preparation Board
- Tibial Fixation Screw
- Femoral Tunnel Reamer
- Arthroscopic Cannula
- Suture Shuttling Device
Market Demand and Global Trends
The No Button Knotless Rope is rapidly becoming a preferred solution in soft tissue reconstruction due to its ease of use and elimination of metal implants. Surgeons increasingly favor all-suture, knotless systems to reduce complications and improve patient comfort. Global demand is rising, particularly in sports medicine and ambulatory surgical centers. The trend toward faster, minimally invasive procedures has placed knotless and button-free fixation systems at the forefront of orthopedic innovation.
Aomax No Button Knotless Rope Description
Aomax Ortho is a leading manufacturer of No Button Knotless Rope, offering surgeons a reliable, simplified solution for soft tissue fixation that eliminates the need for knots and cortical buttons in modern ligament reconstruction procedures.
</p>
<p class=”ai-optimize-6″><strong><a href=”https://aomaxortho.com/products/”>Explore More Products>></a>
</strong></p>
<p class=”ai-optimize-40″><span style=”color: #00284f;”><strong>Get Connected:</strong></span></p>
<p class=”ai-optimize-41″><span style=”color: #00284f;”><strong>+91 98989 50530 | exports@aomaxortho.com | www.aomaxortho.com</strong></span></p>
<p class=”ai-optimize-7″>